A research team led by Dr. Sung Mook Choi from the Korea Institute of Materials Science (KIMS) has made significant strides in hydrogen production by developing a new non-precious metal catalyst. This breakthrough, achieved in collaboration with Professor Seung-Hwa Lee at Changwon National University, could potentially reduce the reliance on expensive precious metals commonly used in hydrogen production technologies.
The team designed a proprietary catalyst specifically for the oxygen evolution reaction (OER) within anion exchange membrane water electrolysis (AEMWE). This new catalyst features a layered structure optimized for alkaline environments, allowing it to operate efficiently without the high costs associated with precious metals.
Addressing Durability Challenges
AEMWE is increasingly recognized as a safe and cost-effective technology for hydrogen production. However, the existing non-precious metal catalysts have struggled with long-term stability in alkaline conditions. Traditional transition metal-based catalysts often face issues such as structural degradation and metal dissolution over prolonged use, which diminish their catalytic effectiveness.
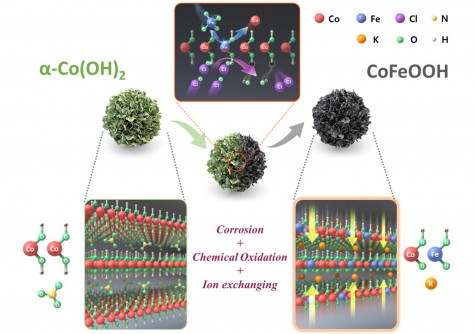
To overcome these challenges, the research team engineered a cobalt (Co) and iron (Fe)-based oxyhydroxide catalyst, referred to as CoFeOOH, with a carefully designed layered structure. This innovative approach allows for better control of the electronic properties of the catalyst’s active surface and optimizes the reaction pathways involved in the oxygen evolution process.
By introducing iron into the CoFeOOH structure, the team effectively modulated the electronic state of the cobalt centers. This adjustment lowered the energy barriers associated with critical adsorption and desorption steps during the reaction, leading to higher current densities even at low overpotentials. Remarkably, the catalyst maintained stable performance without structural degradation during extended operation.
Commercialization Prospects and Future Implications
To further enhance the catalyst’s durability, the researchers developed a unique technique involving controlled chemical oxidation of the catalyst surface. This innovation resulted in a stable surface structure that supports the oxygen evolution reaction in alkaline environments.
The catalyst was successfully applied in a unit cell of AEMWE, confirming its performance and durability under practical operating conditions, surpassing laboratory-scale evaluations. This advancement indicates that non-precious metal OER catalysts can be effectively integrated into AEMWE systems, paving the way for potential commercialization.
Once brought to market, this technology promises to facilitate the development of cost-effective and efficient AEMWE systems, significantly reducing reliance on precious metals. It is also expected to contribute to the growth of clean hydrogen production and improve technological self-sufficiency regarding key materials used in water electrolysis catalysts.
Dr. Sung Mook Choi expressed enthusiasm about the findings, stating, “This research represents a case in which the limitations of non-precious metal–based catalysts were overcome through structural design.” He added, “We will continue our efforts to accelerate the commercialization of green hydrogen production technologies based on anion exchange membrane water electrolysis and contribute to the realization of a hydrogen-based society.”
This research received support from various programs including the National Research Laboratory for Hydrogen (H2 NEXT ROUND) program of the National Research Foundation of Korea (NRF), as well as the institutional research program of KIMS and the Excellent Paper Support Program of Changwon National University. The findings were published online on December 1, 2025, in the prestigious journal ACS Nano, which has an impact factor of 16.1.
The Korea Institute of Materials Science (KIMS) is a government-funded research institute that specializes in materials technologies, contributing significantly to the Korean industry through research and development, testing, and evaluation.







