A team of researchers from the University of Manchester and the University of Oxford has successfully synthesized stable nitrogen chain radical anions under ambient conditions. This significant breakthrough, detailed in a study published in Nature Chemistry on February 28, 2026, marks a pivotal advancement in the understanding of nitrogen chemistry.
Understanding Nitrogen Chain Reactivity
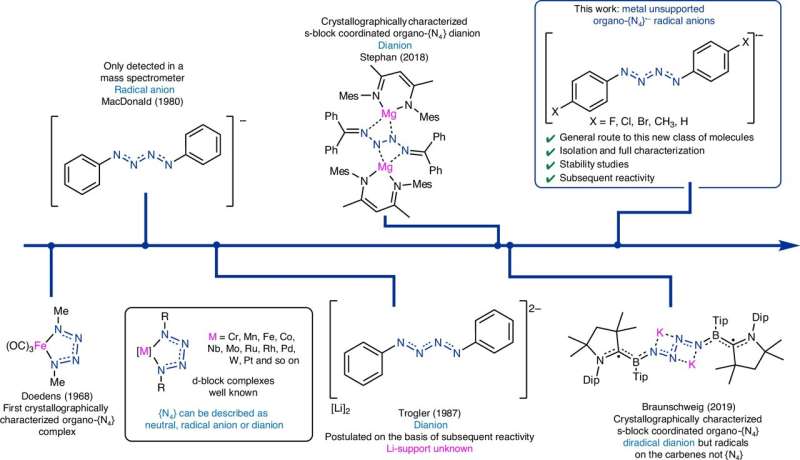
Long-chain nitrogen ions, particularly those with more than three nitrogen atoms, are typically highly reactive and challenging to study. These molecules, often found in Earth’s ionosphere and other planetary atmospheres, usually require extreme conditions for stability, such as ultrahigh pressure. Their rapid decomposition, often resulting in the release of N2, complicates laboratory efforts to isolate and analyze them.
The authors of the study emphasize that nitrogen chains have potential applications in various fields, including propellants, explosives, and gas generators. They note, “Studying nitrogen chain ions under ambient conditions presents a formidable challenge, and a better understanding of their electronic structures should reveal a wealth of hitherto untapped chemical space.”
Previous research efforts have attempted to isolate nitrogen chains, with some success in detecting nitrogen chain ions through mass spectrometry or trapping them at low temperatures. While some studies synthesized metal-coordinated nitrogen chains, these often resulted in altered properties due to the influence of the metal. Organic-substituted N4 chains have been reported in the past but lacked stability and complete characterization.
Achieving Stability Under Ambient Conditions
In their recent study, the researchers isolated five crystalline molecules featuring unsupported tetra-nitrogen radical anion ({N4}•−) chains under normal laboratory conditions. This was accomplished by reducing para-substituted phenyl azides using potassium graphite. Notably, one derivative, [(4-BrC6H4)2N4]•−, remained stable for up to six weeks when stored in anaerobic conditions.
The findings revealed that the radical character was primarily localized along the N4 chain, particularly at the terminal nitrogen atoms. Despite the stability of some derivatives, the team noted that others exhibited instability and could explode upon isolation. They further explored related derivatives, including [(4-FC6H4)2N4]•− and [(Ph)2N4]•−, analyzing variations in spin density and stability due to aromatic substitution.
Reactivity experiments conducted by the team demonstrated that the {N4} chains could cleave into N1 and N3 fragments and serve as sources of nitrene radical anions. These findings indicate that the synthesized molecules may function as storable nitrogen group transfer reagents.
The authors concluded that their results align with the reactivity studies, which showed that the generation of a nitrene radical anion is activated to produce an amide. This research could potentially lead to safer and more efficient applications of nitrogen in various industries, providing storable sources of reactive nitrogen species for chemical synthesis.
As stated by the study authors, “Efforts are now focused on exploring the additional reactivity patterns of {N4}•−-containing molecules, to enhance our understanding of their chemical properties and to fully unlock their potential as gram-scale storable nitrene synthons.”
The implications of this research extend beyond academic inquiry, highlighting the potential for new materials that could revolutionize nitrogen chemistry and its applications in industry.